 The earliest version of the CRT was known as the “Braun tube”, invented by the German physicist Ferdinand Braun in 1897. These rays were named as anode rays or canal rays. In 1886 Goldstein discovered existence of positively charged rays in the discharge tube by using perforated cathode. The discovery of protons can be attributed to Rutherford. During this period, his research resulted in a nuclear reaction which led to the first ‘splitting’ of the atom, where he discovered protons. The proton was discovered by Ernest Rutherford in the early 1900’s. Michael Faraday had consulted Whewell for name ideas for a paper he was writing on electrolysis. 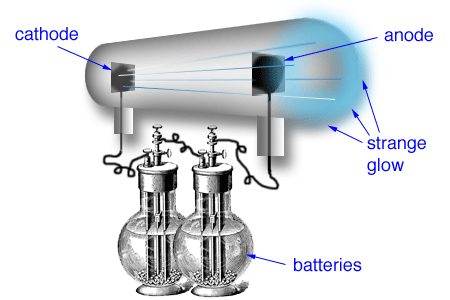
It comes from the Greek word kathodos, which means “way down” or “descent” and refers to the setting sun. The word “cathode” was coined in 1834 by William Whewell. They are produced from the gaseous atoms by knock out of the electrons by high speed cathode rays. What is the origin of cathode ray?Ĭathode rays originate from the cathode whereas anode rays are not produced from the anode. In a device that uses electricity, the cathode is the negatively charged electrode. Why is it called cathode?Ĭathodes get their name from cations (positively charged ions) and anodes from anions (negatively charged ions). 
In 1886 Eugen Goldstein noted that cathode-ray tubes with a perforated cathode emit a glow from the end of the tube near the cathode. See also How long would it take to exit our galaxy? Who discovered cathode rays Goldstein?Įugen Goldstein. The German physicist Karl Ferdinand Braun designed this cathode-ray vacuum tube to visually capture the oscillations in electrical circuits. What is discovered by Goldstein?ĭespite Thomson’s 1897 announcement of subatomic “corpuscles,” he wasn’t the only one hunting for the electron. This proved that the negative charge and the ray were inseparable and intertwined. Thomson’s First Cathode Ray Experiment He found that by applying a magnetic field across the tube, there was no activity recorded by the electrometers and so the charge had been bent away by the magnet. He called his atom the plum pudding model. To explain the neutrality of atoms, Thomson proposed a model of the atom in which negative electrons are scattered throughout a sphere of positive charge. Thomson discovered the first subatomic particle, the electron, while researching cathode rays. It is 100 years since Ernest Rutherford published his results proving the existence of the proton. On his return from America, he achieved the most brilliant work of his life – an original study of cathode rays culminating in the discovery of the electron, which was announced during the course of his evening lecture to the Royal Institution on Friday, April 30, 1897. They were called cathode rays because they were emitted from the cathode of the vacuum tube. See also What Is An Illustration Of Something That Does Not Exist Who named cathode rays?Įugen Goldstein coined the term cathode rays in 1876. In 1838, Michael Faraday applied a high voltage between two metal electrodes at either end of a glass tube that had been partially evacuated of air, and noticed a strange light arc with its beginning at the cathode (negative electrode) and its end at the anode (positive electrode). Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” Who discovered cathode and anode rays? 
Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Who discovered electrons and cathode rays? During his experiment he discovered electrons and it is one of the most important discoveries in the history of physics. Thomson experimenting with cathode ray tubes. The Cathode ray experiment was a result of English physicists named J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
